Clinical record
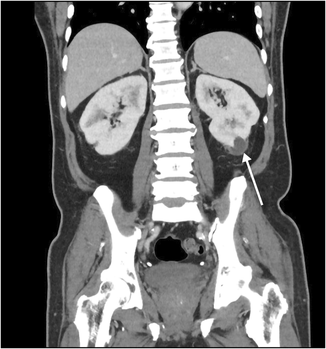
A 65‐year‐old man of Indonesian ethnicity with type 2 diabetes mellitus and hypertension first presented to a peripheral hospital in March 2023 with fevers and diabetic ketoacidosis. He was treated for urosepsis, with Klebsiella pneumoniae isolated in urine and blood cultures. Computed tomography (CT) scan showed multifocal, bilateral thickened perirenal infiltrates, which were discussed at a radiology multidisciplinary meeting and thought to be possibly due to lymphoproliferative disease (Box 1). The infectious diseases team was consulted by the treating medical team; however, as the infectious diseases team was located at the tertiary centre, they could not physically review the patient, and gave advice on antibiotic therapy and duration over the phone. The patient was treated with three days of intravenous (IV) antibiotics and discharged on oral amoxicillin with clavulanic acid, completing a 14‐day antibiotic course. Outpatient screening tests for lymphoproliferative disease and haematology follow‐up were scheduled, with a repeat CT scan planned for three months post‐discharge. Notably, a month prior to his hospital presentation, he travelled to Indonesia, where he required overnight hospital admission for haematemesis.
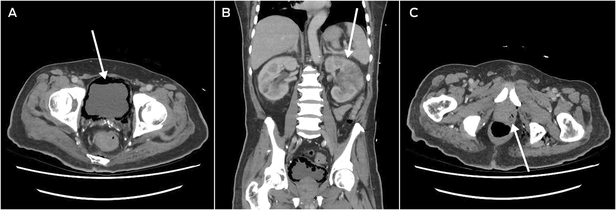
He returned to hospital six weeks after discharge from the Australian hospital, after being found unresponsive at home, with a preceding two‐week history of lethargy, fatigue, and anorexia. He was in septic shock with a temperature of 39°C, tachycardia, tachypnoea, and a Glasgow Coma Scale score of 10. CT scan revealed severe emphysematous cystitis (Box 2, A), left‐sided pyelonephritis (Box 2, B), prostatic abscess (Box 2, C), and gas within the lower pelvis. Operative source control with complete urinary exenteration was deemed not survivable. He was admitted to the intensive care unit, catheterised, and treated with IV meropenem, vancomycin and clindamycin. The infectious diseases team reviewed him within 24 hours of his admission, identifying his epidemiological risk factors for hypervirulent K. pneumoniae. Urine and blood cultures grew K. pneumoniae noted to have a hypermucoviscous phenotype (positive string test), following which his antibiotic regimen was rationalised to high dose IV ceftriaxone monotherapy. After initial clinical improvements, his condition deteriorated with new fevers, worsening confusion, and rising inflammatory markers and lactate on his blood tests, and IV metronidazole and ciprofloxacin were added. He gradually improved, receiving a further six weeks of oral ciprofloxacin monotherapy.
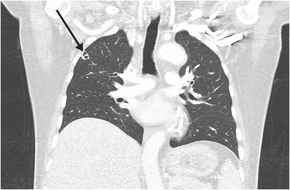
His clinical course was complicated by septic thromboembolism, including left renal vein thrombosis, multi‐territorial subcortical strokes, and disseminated lung infection, with multiple peripheral cavitating nodules (Box 3). Transoesophageal echocardiogram and cardiac magnetic resonance imaging showed no evidence of infective endocarditis or patent foramen ovale.
Following an extended rehabilitation period due to critical illness myopathy, he was discharged home after a four‐month hospital admission.
Discussion
Classical K. pneumoniae is an encapsulated Enterobacterales gram‐negative bacillus, and a common cause of community and health care associated urinary tract infections.1,2,3,4,5 Severe classical K. pneumoniae infection most commonly occurs in hosts with significant comorbidities or immunocompromise, and may involve the lungs, abdominal cavity, and urinary system.1,2,3,4,5
Hypervirulent K. pneumoniae infection was first reported in Taiwan in the mid‐1980s, with pyogenic liver abscesses developing in the community in otherwise healthy hosts.2,4,5 Subsequent reports of hypervirulent K. pneumoniae infection are mainly from east Asia but have been described worldwide, especially in people of Asian ethnicity and geographic origin.4 In Australia, the incidence and transmission are unknown, as reports are from case studies only.1,2 Most Australian hypervirulent K. pneumoniae infections have hosts of Asian ethnicity or history of travel to Asia. Studies within endemic regions suggests faecal–oral transmission as the route for person‐to‐person spread.1 Responsible strains are also referred to as hypermucoviscous or hypermucoid K. pneumoniae because of their hypermucoid appearance on agar plates. Hypermucoviscosity is identified via a string test; the test is considered positive if the bacterial colony can be stretched further than 5 mm from the agar plate with an inoculation loop.1,3 Hypervirulent K. pneumoniae virulence factors include capsular serotypes (K1 and K2), a large virulence plasmid (pLVPK) that encodes aerobactin, salmochelin and rmpA, as well as genetic clonal complex 23 and sequence type 23.2 Although these virulence factors increase the ability of hypervirulent K. pneumoniae to metastasise, there are few known patient risk factors or predictors for metastatic disease.2 Type 2 diabetes mellitus has been implicated in patients with hypervirulent K. pneumoniae2 and generally predisposes patients to bacterial infection; however, patients without diabetes or other comorbidities are still at risk of life‐threatening disease.
Patients with hypervirulent K. pneumoniae infection may be relatively young but frequently present with severe disease. Common symptoms include fevers, nausea, vomiting, and abdominal pain.1 Disseminated extrahepatic hypervirulent K. pneumoniae infection is seen in up to one‐third of patients. Common sites of seeded infection include the lungs, eyes, or central nervous system, commonly with concomitant liver abscess.1 High rates of thrombotic complications and one case of infective endocarditis have been reported.1 While the initially described pathognomonic syndrome was primary pyogenic liver abscess with or without disseminated infection, primary extrahepatic infection is now widely reported with sites including the urinary tract, biliary tract, osteoarticular foci, and soft tissue.1,4
Hypervirulent K. pneumoniae infection frequently causes severe morbidity, and mortality rates may be up to 35% with bacteraemia.1 Markers for increased mortality risk include gastrointestinal fistula, widely metastatic infection, septic shock, acute respiratory failure, and gas formation on imaging.1 The longer term sequelae can be devastating, with 70% of patients with ocular or central nervous system metastatic disease experiencing permanent disability.1
Optimal treatment of hypervirulent K. pneumoniae infection includes adequate source control by abscess drainage and infected tissue debridement, and extended antibiotic therapy, generally between two and six weeks depending on site and extent of infection and adequacy of source control.1 The incidence of multidrug‐resistant hypervirulent K. pneumoniae is increasing,1,4,5 with carbapenem resistance of particular concern, risking this becoming a progressively difficult infection to treat. Infectious diseases specialists should be consulted early to direct investigations and antibiotic therapy. Currently, there are no published guidelines or evidence for screening for metastatic disease; however, infectious diseases physicians may advocate for neuroimaging or ophthalmic review.
This case highlights the devastating consequences of hypervirulent K. pneumoniae infection. Early inpatient review by the infectious diseases team may have flagged this patient's epidemiological risk factors during his first admission, and the case highlights the importance of taking a thorough history. While primarily endemic within Asia, global prevalence is increasing.1 Given Australia's multi‐ethnic population and proximity to Asia, clinicians should be alert to this life‐threatening infection.
Lessons from practice
- Hypervirulent Klebsiella pneumoniae can cause severe, life‐threatening infections in both healthy and immunocompromised hosts, in both community and hospital settings.
- Patients can present with non‐specific signs of infection, with high rates of mortality and morbidity, particularly in patients with metastatic infection.
- Primary sites of infection can vary and may include the urinary tract in addition to the more commonly recognised liver abscess and other metastatic organ foci.
- Clinical suspicion with early involvement of infectious diseases specialists is recommended to aid with appropriate identification and antimicrobial treatment.
Box 1 – Computed tomography scan (coronal plane) showing thickened perirenal infiltrates around the kidney (arrow)
Provenance: Not commissioned; externally peer reviewed.
- 1. Choby J, Howard‐Anderson J, Weiss D. Hypervirulent Klebsiella pneumoniae – clinical and molecular perspectives. J Intern Med 2020; 287: 283‐300.
- 2. Lee C, Lee J, Park K, et. al. Antimicrobial resistance of hypervirulent Klebsiella pneumoniae: epidemiology, hypervirulence‐associated determinants, and resistance mechanisms. Front Cell Infect Microbiol 2017; 483: 1‐13.
- 3. Russo T, Marr C. Hypervirulent Klebsiella pneumoniae. Clin Microbiol Rev 2019; 32: e00001‐19.
- 4. Shon A, Bajwa R, Russo T. Hypervirulent (hypermucoviscous) Klebsiella pneumoniae. A new and dangerous breed. Virulence 2013; 4: 107‐118.
- 5. Russo T, Olson R, Fang C, et. al. Identification of biomarkers for differentiation of hypervirulent Klebsiella pneumoniae from classical Klebsiella pneumoniae. J Clin Microbiol 2018; 56: e00776‐18.






Open access:
Open access publishing facilitated by The University of Western Australia, as part of the Wiley ‐ The University of Western Australia agreement via the Council of Australian University Librarians.
Patient consent:
The patient provided written consent for publication.
No relevant disclosures.